Personalizing Perfusion: Hemodynamic Management in Pediatric Anesthesia
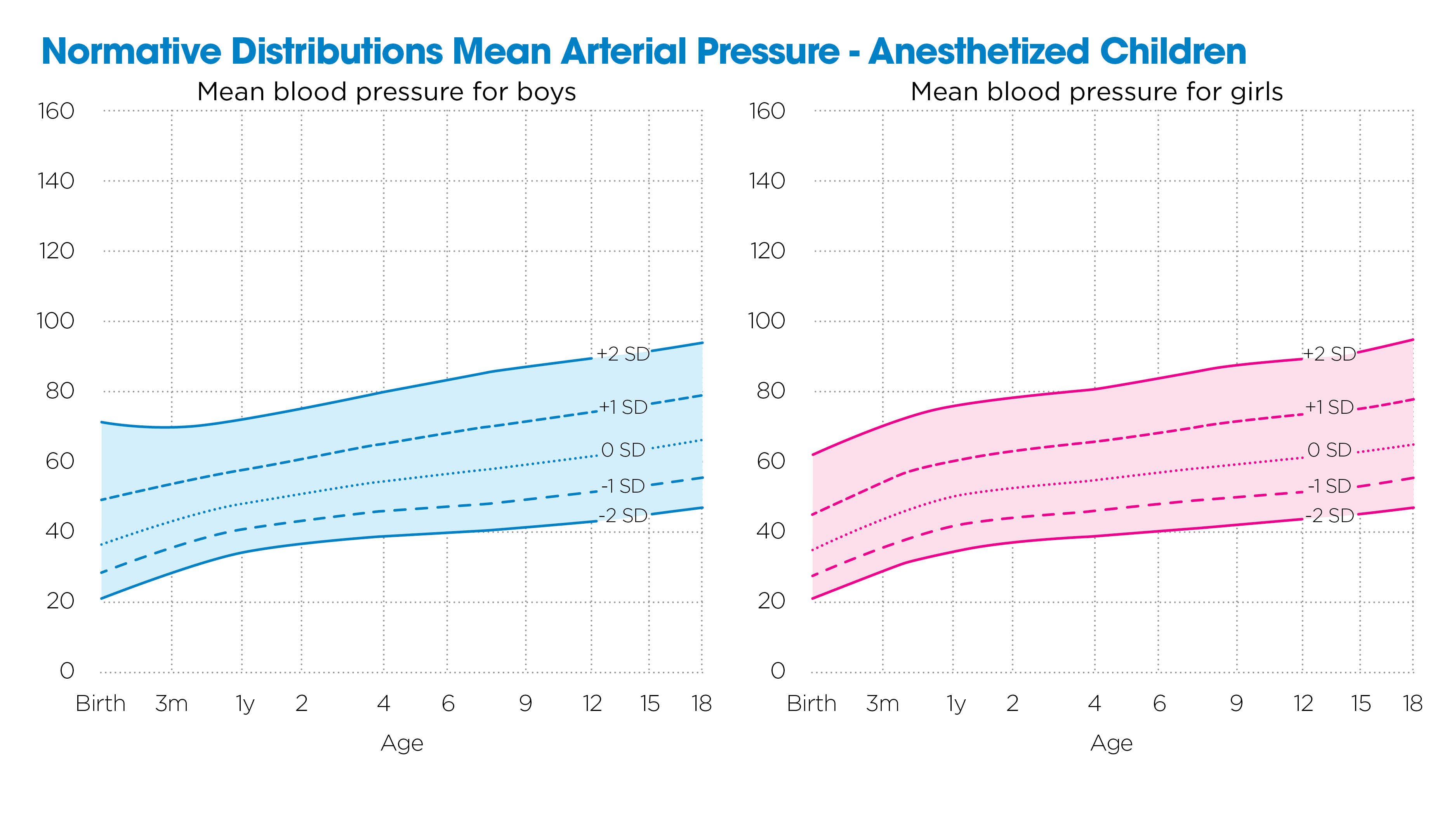
Ensuring adequate oxygen delivery to the brain and vital organs is among the most critical responsibilities of the pediatric anesthesiologist. Hemodynamic changes are common during anesthesia and relate to the effects of medications, ventilation, positioning and the surgical procedure, including blood and fluid loss. Strategies to optimize hemodynamic parameters can be guided by theoretical or experimental models, population norms and personalized measures. Traditionally, we’ve relied on fuzzy logic, adjusting interventions based on population blood pressure norms and a child’s pre-anesthesia baseline.¹ While this approach is adequate for low-risk patients and procedures, it does not provide enough information to guide interventions in the complex care commonly provided at Children’s Wisconsin.
Near-infrared spectroscopy (NIRS) allows us to observe organ-specific hemodynamic changes in real time using continuous, noninvasive optical probes placed on the skin over vital organs. Because organs lie close to the surface in smaller bodies, NIRS is especially useful in neonates, infants and young children, in whom invasive techniques are more challenging and risky.² With forehead and flank probes, we can track organ-level perfusion in the brain and kidney throughout the perioperative period.
Across a range of patient populations, we’ve observed differential cerebral and renal control mechanisms and responses to physiologic stress not apparent with blood pressure alone. Because normal physiologic function involves dynamic changes in regional blood flow, trade-offs between organs are expected but not always desirable. In children, perfusion can drop to critical levels even when mean arterial blood pressure (MABP) appears normal. Organ-specific monitoring reveals this in real time in ways that traditional metrics cannot.³
To interpret these signals meaningfully, we’ve built multidimensional models using hundreds of thousands of NIRS data points, many from high-risk neonates with congenital heart disease. These models now guide real-time clinical decisions, even when using noninvasive methods. We use them to determine when to give fluids, initiate vasopressors or adjust ventilatory settings.
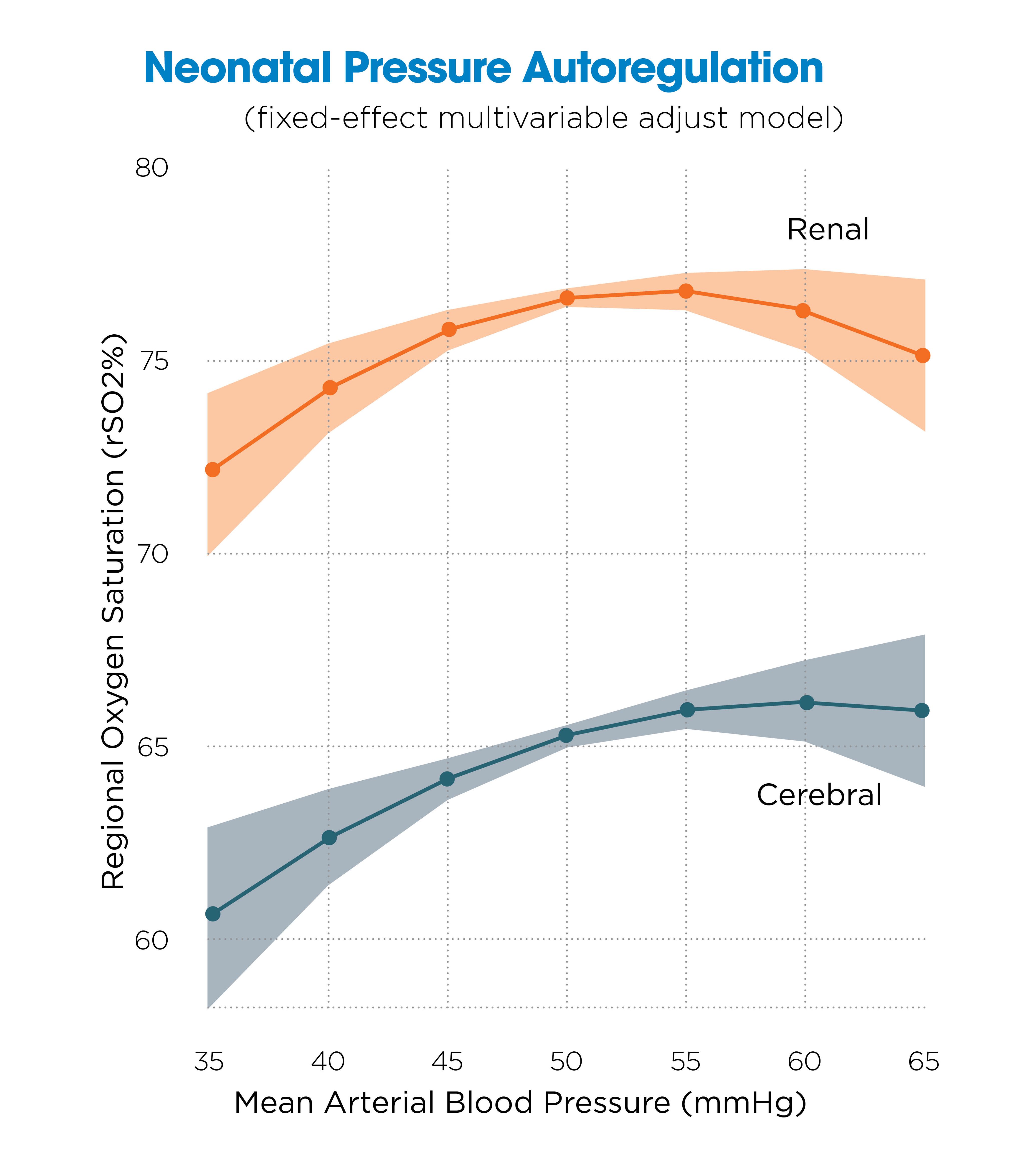
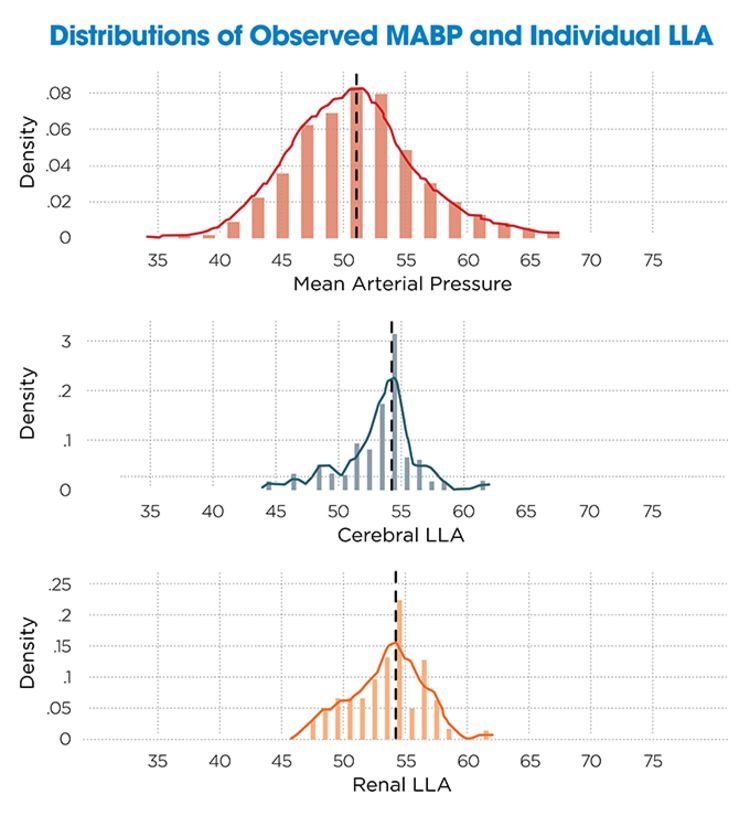
In a study of 200 neonates, we evaluated the autoregulation relationship between MABP and oxygen saturation in the brain (rSO₂C) and kidneys (rSO₂R) over a 48-hour perioperative period.⁴ Using nonlinear regression, we identified the lower limit of autoregulation (LLA) — the threshold below which the organ can no longer maintain stable perfusion. We found most LLAs in the 45–50 mmHg range for both regions, but cerebral perfusion dropped more steeply as pressure declined. Interestingly, renal saturation also decreased at pressures above 65 mmHg, suggesting that some increases in blood pressure come at the expense of organ perfusion.
These findings are clinically meaningful. In our population, MABP fell below the LLA in about 25% of patients, and 76% experienced at least one hour of hemodynamic vulnerability. Based on population data, standard reference ranges often miss these high-risk periods. Most LLAs fell near the 75th percentile of conventional anesthetized blood pressure norms, reinforcing the need for individualized thresholds.
Broad application of this noninvasive technology lets us observe hemodynamic responses otherwise invisible until the onset of organ dysfunction. NIRS provides visibility into organ-level perfusion and allows early intervention, even when traditional metrics look acceptable. At Children’s Wisconsin, this two-site NIRS monitoring strategy has become standard in high-risk care.
Augmented hemodynamic monitoring with both pressure and oximetric techniques gives us a deeper understanding of how children respond to anesthesia, surgical stress and critical illness. With insights from this monitoring strategy, we can deliver personalized anesthesia care that more closely targets each patient’s individual needs.
References
1. De Graaff JC, Pasma W, Van Buuren S, et al. Reference Values for Noninvasive Blood Pressure in Children during Anesthesia: A Multicentered Retrospective Observational Cohort Study. Anesthesiology. 2016;125(5):904–13.
2. Hoffman GM, Tweddell JS. Regional blood flow monitoring in the perioperative period. In: Kleinman CS, Seri I, editors. Hemodynamics and Cardiology. Philadelphia: W.B. Saunders; 2012. p. 473–87.
3. Scott JP, Hoffman GM. Near-infrared spectroscopy: Exposing the dark (venous) side of the circulation. Paediatr Anaesth. 2014;24(1):74–88.
4. Hoffman GM, Tanem JM, Scott JP, et al. Pressure autoregulation in cerebral and renal beds in neonates following deep hypothermic cardiopulmonary bypass. ASA 2024: A1157. Philadelphia, PA; 2024.